Arvinas + Pfizer License VEPPANU to Rigel — $85M Upfront, $320M Milestones
Arvinas and Pfizer's May 12 8-K licenses FDA-approved breast cancer PROTAC VEPPANU to Rigel for $85M upfront and up to $320M milestones plus tiered royalties. Here's what the deal restructures.
An FDA-approved breast cancer drug just changed hands without a merger. On May 12, 2026, Arvinas, Inc. (NASDAQ: ARVN) filed an 8-K disclosing that it and Pfizer Inc. (NYSE: PFE) have entered into a license agreement with Rigel Pharmaceuticals, Inc. (NASDAQ: RIGL) for the exclusive global development, manufacturing, and commercialization rights to VEPPANU (vepdegestrant), an FDA-approved PROTAC for breast cancer. Headline economics: $70 million upfront, $15 million on completion of transition activities, up to $320 million in milestones, plus tiered royalties.
The structure of the deal is more informative than the dollar amounts. Arvinas and Pfizer co-developed VEPPANU under a longstanding collaboration. Licensing the commercialized asset out to a third party — Rigel, a smaller specialty biotech — is the kind of disclosure that signals a co-development partnership has decided the asset is worth more outside its current corporate home than inside it.
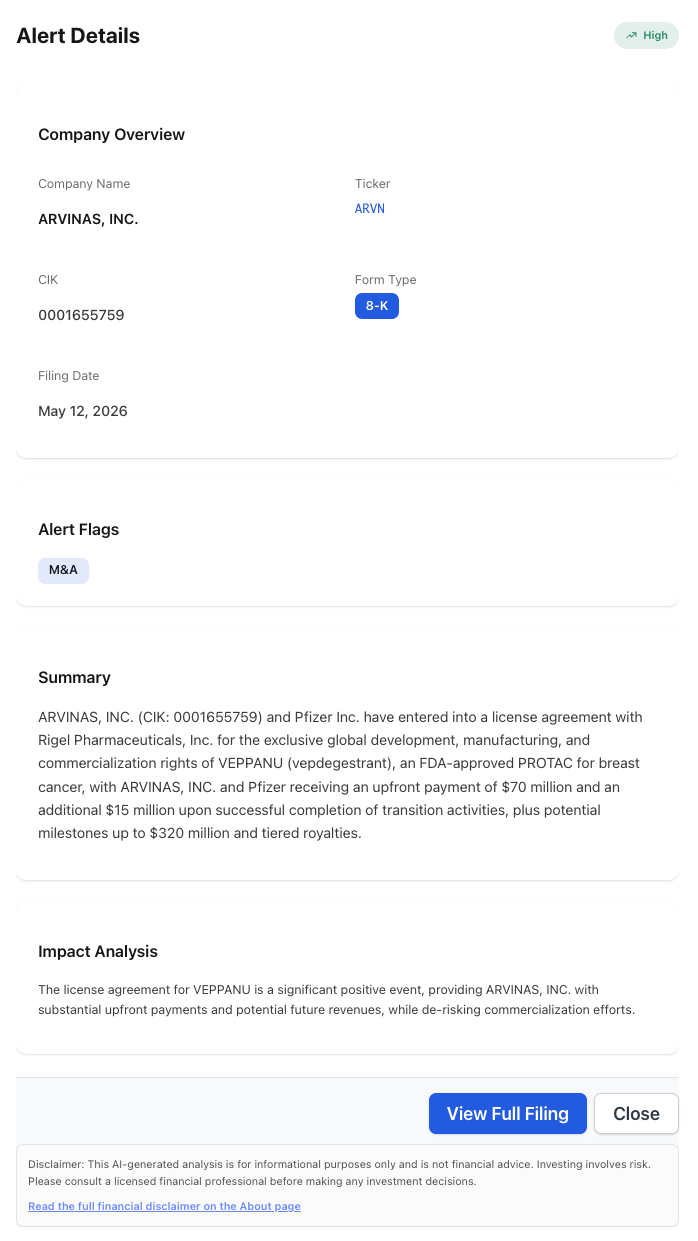
The M&A flag firing on a licensing transaction is the diagnostic. A pure research collaboration would carry an “other” or partnership tag; this filing is being read by the system as a structural asset transfer, which is exactly what an exclusive global rights handoff of a commercial-stage product is. The Impact Analysis line — “de-risking commercialization efforts” — is the part most readers will skim past. It is the most honest sentence in the summary.
What the Filing Discloses
The 8-K’s deal-economics line items match the press release verbatim. That alignment is itself a useful signal: the AI extraction layer is pulling structured terms out of the filing, not paraphrasing the headline:
- Asset: VEPPANU (vepdegestrant), an FDA-approved PROTAC (proteolysis targeting chimera) for breast cancer.
- Counterparty: Rigel Pharmaceuticals, Inc. (
RIGL), acquiring exclusive global development, manufacturing, and commercialization rights. - Upfront payment to Arvinas + Pfizer: $70 million.
- Transition payment: $15 million upon successful completion of transition activities.
- Potential milestones: up to $320 million.
- Royalties: tiered, structure not disclosed at the filing level.
- Severity classification: High / Opportunity for
ARVN. Flag: Merger/Acquisition.
That last classification matters. The Merger/Acquisition flag fired on a licensing deal, not a corporate combination, because the filing parser is reading the structural transaction: an exclusive global rights transfer of a commercialized asset is economically closer to an asset purchase than to a research collaboration. The flag stack is calibrated for the substance of the disclosure, not the legal form.
$85M Upfront Is the Number You Recognize, But Not the Number That Matters
The headline number being repeated across coverage is $85 million — the sum of the $70M upfront and the $15M transition payment. That number is mostly a near-term cash event for Arvinas and Pfizer.
The structurally important numbers are the $320 million in milestones plus tiered royalties. Here is why:
- Milestones are pipeline derivatives. A $320M ceiling on milestones implies that Arvinas and Pfizer projected meaningful additional regulatory, commercial, and sales-based achievements for VEPPANU. The size of the milestone pool is the part of the deal that says “we still think this asset has runway.”
- Tiered royalties are the perpetuity. Tiered royalties on commercialized sales create a long-tail revenue stream for Arvinas and Pfizer that scales with VEPPANU’s commercial success. Without disclosed tiering, the headline economics underestimate the deal’s lifetime value.
- The upfront is not the valuation. In biotech licensing, the upfront is the entry price. The milestone pool plus royalties is where the actual economics sit, often representing 5-10x the upfront over the asset’s lifecycle in successful cases.
For comparison: a $70 million upfront on a pre-approval PROTAC asset would be a meaningful biotech licensing deal. A $70 million upfront on an FDA-approved commercial-stage PROTAC is a strikingly modest entry price — which is what the milestone-and-royalty back-end is built to compensate for.
Why Rigel Is the Acquirer Here
Rigel Pharmaceuticals is a specialty biotech with a commercial hematology and oncology portfolio. Picking up VEPPANU global rights is not the kind of transaction Rigel does as a routine matter — it is the kind of transaction that doubles or triples the size of a small biotech’s commercial portfolio in one filing.
Three things the deal structure tells you about Rigel’s position:
- Rigel has commercial infrastructure. Acquiring exclusive global development, manufacturing, AND commercialization rights is only feasible for an acquirer that can actually commercialize. Rigel’s existing oncology footprint is the operational base that makes this deal possible.
- Rigel is paying back-loaded. $85M upfront for a commercial-stage FDA-approved asset is a structure that favors a smaller acquirer’s balance sheet. The bigger payments are conditional, which means Rigel is taking on execution risk rather than upfront balance-sheet risk.
- Rigel is replacing pipeline. This is the second material pipeline event Rigel has been involved in this quarter. On April 25, 2026, Eli Lilly terminated its CNS collaboration with Rigel (covered here). Picking up VEPPANU global rights is the offsetting capability acquisition.
That last point is the cross-filing pattern. Rigel losing the Lilly collaboration on RIPK1 and gaining VEPPANU global rights within a 17-day window is not a coincidence — it is the visible footprint of an active business-development team reshaping the pipeline in real time. The filings are how you see that reshaping happen.
Why Pfizer Is the Quiet Half of This Deal
The 8-K is filed by Arvinas because the licensing transaction triggered Arvinas’s disclosure obligations. Pfizer is the co-licensor and will share the economics, but Pfizer’s quarterly revenue base is large enough that VEPPANU’s economics don’t trigger an immediate 8-K disclosure on Pfizer’s side.
For investors watching PFE, the relevant filings are:
- Pfizer’s next 10-Q — should disclose VEPPANU licensing income as part of the collaboration revenue line item, with the upfront recognized over the relevant performance obligation period.
- Pfizer’s next earnings 8-K — likely references the deal in the prepared remarks or Q&A, especially if it materially shifts the company’s oncology partnership economics for the quarter.
- Any subsequent Schedule 13G filings — if Rigel issues equity to fund the milestone or royalty obligations, the cap-table flow becomes the next signal.
The bilateral signal in this deal runs across three tickers, not two. ARVN files the 8-K. PFE collects part of the economics. RIGL acquires the asset and the future commercial risk. Three different filing tracks generate three different alert flows for the same underlying transaction.
The PROTAC Context
VEPPANU is a PROTAC — a protein degrader, not a traditional small molecule. The mechanism is one of the most-watched modalities in oncology drug development:
- PROTACs use the cell’s own ubiquitin-proteasome system to degrade target proteins rather than inhibit them
- FDA approval of a PROTAC is itself a meaningful regulatory milestone, given that the modality has historically been clinical-stage
- VEPPANU (vepdegestrant) targets the estrogen receptor in breast cancer — a clinically validated target with substantial commercial precedent
Arvinas and Pfizer’s joint development of VEPPANU represented one of the highest-profile PROTAC programs in oncology. Licensing the commercial rights out — while presumably retaining some ongoing development obligations under the collaboration framework — is a strategic decision about portfolio focus, not a sunset on the modality.
The most informative biotech licensing 8-Ks are the ones where a co-developed, commercial-stage asset gets handed to a third party. That structure tells you something the upfront number can’t.
What This Looks Like on a Watchlist
A watchlist tracking the oncology PROTAC space across ARVN, PFE, and RIGL would have surfaced the May 12 8-K as the inflection event. The flag stack:
- ARVN — High / Opportunity, flag: Merger/Acquisition, summary: $70M upfront, $15M transition, up to $320M milestones plus royalties on VEPPANU.
- PFE — Cross-listed alert via the named counterparty linkage. Subscribers watching
PFEsee the deal even though Pfizer is not the filing entity. - RIGL — Cross-listed alert as the receiving counterparty. For Rigel investors, this is the most material filing of the quarter and the offsetting positive to the Lilly RIPK1 termination 17 days earlier.
That tri-lateral linkage is the architecture biotech investors actually need. Pipeline deals rarely involve only two parties — co-development collaborations, royalty trusts, and milestone-bearing arrangements often distribute economics across three or more named entities. Single-ticker monitoring catches one and misses the rest.
The Cross-Filing Pattern: Rigel’s Quarter
Pulling the Rigel filing track into one view shows the business-development shape clearly:
- April 25, 2026 — Eli Lilly terminates RIPK1 collaboration. Rigel loses a CNS/neuroinflammation pipeline asset. Negative pipeline event.
- May 12, 2026 — Rigel acquires global VEPPANU rights from Arvinas + Pfizer. Rigel adds an FDA-approved commercial oncology asset. Positive pipeline event.
Net pipeline impact in 17 days: from a clinical-stage CNS asset with a major-pharma partner backing out, to a commercial-stage oncology asset with global rights and the partner economics back-loaded. That is a meaningful trade for a specialty biotech, and it is only visible if the monitoring system shows both filings in the same timeline.
NexusAlert’s Investor Trends view for RIGL will plot both events on the same timeline — termination 8-K on April 25, licensing 8-K on May 12 — with the AI-derived sentiment trajectory captured across the pair.
How to Track Biotech Licensing Without Reading Every 8-K
Biotech licensing deals are filed as 8-K Item 1.01 disclosures and accompanied by exhibit-level redacted license agreements. The structural deal economics — upfront, milestones, royalties, exclusivity scope — sit in the exhibits and the filed body, not in the press release boilerplate.
NexusAlert’s biotech filing pipeline runs the screen automatically:
- Parses 8-K Item 1.01 disclosures for license-agreement language and dollar magnitudes
- Cross-references named counterparties on multi-party collaborations for tri-lateral alert routing
- Flags FDA-approved-asset transfers as a distinct signal class from pre-approval licensing
- Surfaces milestone-and-royalty structures as separate economic lines from upfront payments
- Combines the flag stack with cross-filing patterns at the company level — so subsequent positive or negative pipeline events surface as related, not isolated
The Arvinas / Pfizer / Rigel licensing 8-K fired as a High-severity Opportunity alert in real time, with deal economics ($70M / $15M / $320M / tiered royalties) parsed into the summary before the headline cycle moved past the $85M number.
Catch the Next Biotech Licensing Deal the Day It Files
Create a free NexusAlert account to get AI-powered alerts on biotech 8-Ks, licensing-agreement disclosures, milestone-and-royalty structures, and multi-party collaboration filings across every public biotech — with deal terms, cross-ticker linkages, and pipeline-event trajectories parsed in real time.
Sources
- Arvinas and Pfizer Enter into a Transaction with Rigel Pharmaceuticals for the Exclusive Global Rights of VEPPANU (vepdegestrant) — GlobeNewswire / Manila Times
- Arvinas, Pfizer sell rights to VEPPANU to Rigel for $85 million — Investing.com
- Arvinas, Pfizer license VEPPANU rights to Rigel — StockTitan
- Eli Lilly Just Killed Its Rigel CNS Collaboration — NexusAlert